 Nivolumab (Opdivo) injection has received FDA approval for treating patients with locally advanced or metastatic urothelial carcinoma (mUC) who have disease progression during or after platinum-containing chemotherapy or disease progression within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. In the CheckMate-275 phase 2 open-label trial, 53 (19.6%) of 270 of patients responded to treatment with nivolumab: 7 patients (2.6%) had a complete response and 46 patients (17%) had a partial response, according to a press release from Bristol-Myers Squibb Company, the maker of nivolumab. Among responders, the median time to response was 1.9 months and the median duration of response was 10.3 months. In the trial, patients received nivolumab 3 mg/kg intravenously every 2 weeks until disease progression or unacceptable toxicity. The recommended dose for mUC is 240 mg administered as an intravenous infusion over 60 minutes every 2 weeks until disease progression or unacceptable toxicity. The primary endpoint was objective response rate, as defined by an independent radiographic review committee.
0 Comments
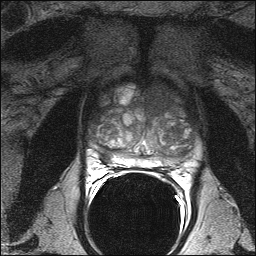 Multiparametric magnetic resonance imaging (MP-MRI) can clarify which men with an elevated PSA (up to 15 ng/mL within 3 months) can safely avoid an immediate prostate biopsy and improve the accuracy of prostate cancer (PCa) diagnosis, according to a new study. In the blinded PROMIS trial (NCT01292291), Hashim U. Ahmed, FRCS, PhD, BM, BCh (Oxon), of the University College London Medical School in the United Kingdom (UK), and colleagues compared findings from 1.5 Tesla MP-MRI biopsy with transrectal ultrasound-guided (TRUS) biopsy against transperineal template prostate mapping (TPM) biopsy as a reference. The investigators posited that TPM is more accurate than TRUS-biopsy as a reference because it samples the entire prostate every 5 mm with an estimated 95% sensitivity for clinically significant PCa, and it is less prone to selection bias than radical prostatectomy specimens. Clinically significant cancers were defined as Gleason score 4 + 3 or above or a maximum cancer core length of 6 mm or longer. Of 576 biopsy-naïve men from 11 centers in the UK, 71% had cancer based on standard TRUS biopsy (10 to 12 cores) with 40% of cases deemed clinically significant. MP-MRI proved more sensitive at finding significant cancers than TRUS biopsy (93% vs 48%), but was less specific (41% vs 96%). MP-MRI elucidates prostate volume, cellularity, and vascularity. “The lower specificity and positive predictive value of MP-MRI means that a biopsy is still required with a suspicious MP-MRI,” Dr Ahmen and colleagues explained in a paper published online in the Lancet. “Men with suspicious MP-MRI areas can have biopsies guided by these findings. Overall, this strategy could improve detection of clinically important prostate cancer and reduce the number of men diagnosed with clinically unimportant disease.” Imaging men first with MP-MRI might avoid biopsy in 27% of men and prevent 5% of overdiagnoses, the investigators estimated. In addition, when TRUS biopsies are guided by MP-MRI findings, up to 18% more clinically significant cancers might be detected than with TRUS biopsy alone. Among the study's limitations, men with prostates greater than 10 mL were excluded from the trial to accommodate the use of TPM. In addition, TRUS biopsy was performed after TPM biopsy, so it is possible that swelling, distortion, and tissue disruption may have occurred and affected results. This study did not target MR-suspicious lesions so it cannot comment on the utility of targeted biopsy. A cost analysis of using MP-MRI in this context is forthcoming. With regard to adverse effects, 5.9% of all recruited patients reported serious adverse events, including 8 cases of sepsis following urinary tract infection. |
Σχετικά
Με το ιστολόγιο αυτό επιχειρείται η κατά το δυνατόν άμεση ενημέρωση σε θέματα που αφορούν ουρολογικές εξελίξεις και δίνεται η δυνατότητα σχολιασμού των αναρτήσεων. Κατηγορίες
All
|
|
Ιατρείο Λαγκαδά: Δ. Μήλιου 24 (έναντι 1ου δημοτικού), Λαγκαδάς
Τ: 2394 020 780, Κ: 693 63 23 794 |
Όροι Χρήσης | Επικοινωνία | Copyright © 2013
 RSS Feed
RSS Feed